First UK private hospital use Speedboat for colorectal cancer case
The London Clinic Performs First-Ever Speedboat Submucosal Dissection for Early Colorectal Cancer

The London Clinic is proud to announce a significant milestone in advanced endoscopic care: Dr Roser Vega has successfully performed the hospital’s first-ever Speedboat submucosal dissection (SSD), offering a highly innovative, organ-sparing treatment for patients with early colorectal cancer.
This achievement marks The London Clinic as the first private hospital in the UK to introduce and deliver this cutting-edge technique, reinforcing its commitment to pioneering minimally invasive treatments and improving patient outcomes.
Speedboat submucosal dissection (SSD) is a form of endoscopic submucosal dissection (ESD) performed using the Speedboat® device, an advanced surgical instrument that combines cutting, coagulation, and dissection capabilities in a single tool. The device uses innovative technology developed and manufactured by Creo Medical, enabling clinicians to perform highly precise and controlled removal of early-stage cancerous and pre-cancerous lesions from the gastrointestinal tract.
Unlike traditional surgical approaches, which may require partial removal of the colon, SSD offers a less invasive, organ-preserving alternative. Patients benefit from reduced recovery times, fewer complications, and the potential to avoid major surgery altogether.
Dr Roser Vega, Consultant Gastroenterologist at The London Clinic, commented:
"We are delighted to bring this advanced technique to patients in the private sector. Speedboat submucosal dissection represents a major step forward in the treatment of early colorectal cancer, allowing us to remove lesions effectively while preserving organ function. This approach significantly improves patient experience and outcomes."
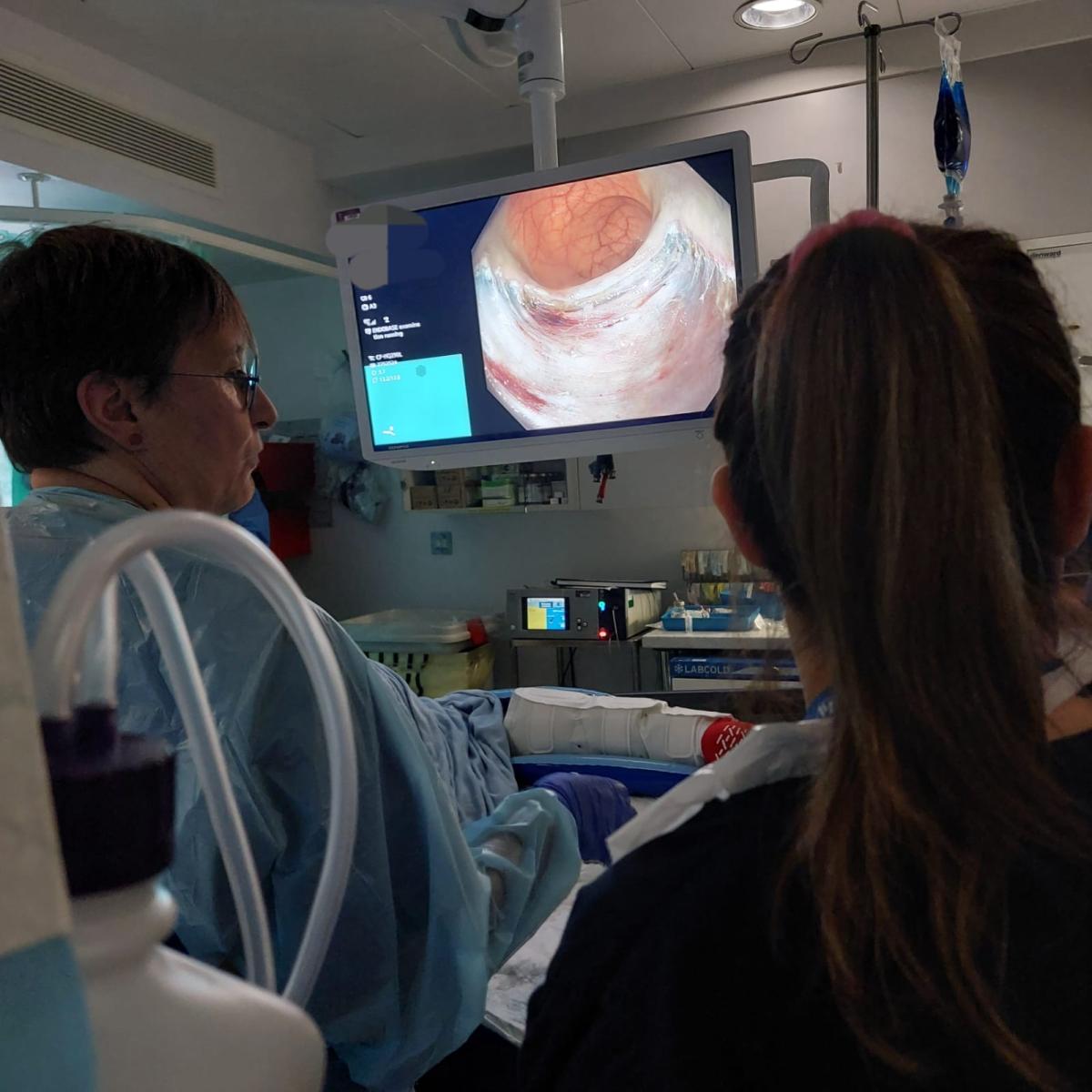
The introduction of SSD at The London Clinic reflects the hospital’s ongoing investment in innovative technologies and specialist expertise. By offering this service, The London Clinic is expanding access to state-of-the-art, minimally invasive cancer treatments within the UK’s private healthcare sector. This milestone further strengthens The London Clinic’s position as a leader in advanced endoscopy and patient-centred care.
The introduction of Speedboat submucosal dissection at The London Clinic reflects continued investment by leading institutions in advanced endoscopic techniques and reinforces Creo Medical’s position at the forefront of minimally invasive cancer care. The procedure further expands access to state‑of‑the‑art endoscopic procedures within the UK, supporting broader adoption across both public and private healthcare systems.
This milestone builds on the growing clinical momentum for Speedboat across key geographies and high‑value procedural areas, including complex upper and lower gastrointestinal interventions, and further validates the scalability and relevance of Creo’s Advanced Energy platform.
Craig Gulliford, Chief Executive Officer of Creo Medical, commented:
“The successful launch of the Speedboat service at The London Clinic is a significant endorsement of our technology and its role in delivering organ‑sparing, minimally invasive cancer treatments in a private healthcare setting here in the UK. Adoption by a leading private hospital highlights the growing clinical confidence in Speedboat, reinforcing our belief that advanced endoscopic products like ours will play an increasingly important role in improving both the clinical outcomes for patients and the accessibility of these procedures. We are delighted to see Speedboat continuing to expand its footprint across centres of excellence and delivering meaningful benefits for both clinicians and patients.”
Posted 29/04/2026
For press enquiries please contact media@creomedical.com. For all other enquiries please visit our Contact page.

