Our Journey
2003

2003
June 2003 - MicroOncology is founded
MicroOncology is founded by Professor Chris Hancock with a vision of improving patient outcomes across indications by bringing advanced energy to therapeutic endoscopy.
2010
2010
MicroOncology rebrands as Creo Medical Group and the company’s focus shifts to bringing advanced energy medical devices to market.
2012

2012
Craig Gulliford is appointed CEO of Creo Medical Group.
2016

2016
Creo Medical is listed on the London Stock Exchange.
2017

2017
Creo Medical Moves into Chepstow HQ.
First patient treated with Speedboat Inject.
CE Mark clearance gained for Speedboat Inject and the CROMA Advanced Energy Platform.
2018

2018
£48.5M raised through share placings to accelerate commercialisation.
First use of Speedboat Inject in mainland Europe and South Africa.
2019

2019
£51.9m raised through share placings to accelerate US programme and progress research and development.
First use of Speedboat Inject in the US and India, with first commercial orders received.
2020

2020
MicroBlate Fine’s first in-human use.
Acquisition of Albyn Medical S.L, Boucart Medical and Endotechnik, providing Creo with established European distribution network and wider portfolio of products.
CE Mark clearance achieved for five new devices, with FDA clearance also gained for SlypSeal Flex and Microblate Fine.
2021

2021
US FDA clearance for MicroBlate Flex.
Opening of US HQ in Danbury.
Creo aquires Aber Electronics Limited – enhancing additional expertise to Creo and a key element of supply chain.
2022

2022
Launch international Pioneer Clinical Education Programme.
Long term collaboration announced with Intuitive, with heads of terms announced to grant further third-party access to Creo’s advanced energy technology.
Creo Medical opens APAC regional hub in Singapore.
2023

2023
First European Speedboat® UltraSlim procedure marks early commercial launch.
2024
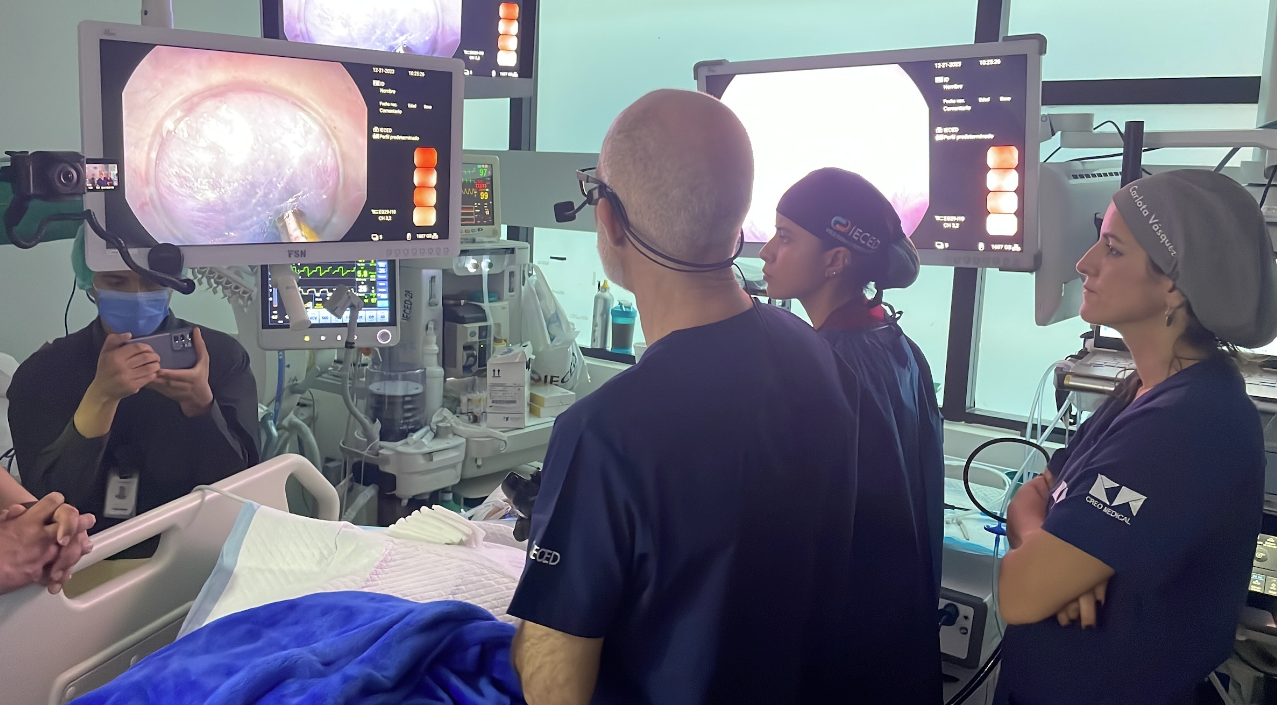
2024
Commercial launch of Speedboat UltraSlim, following a successful limited market release in December 2023, expanding the procedures that can be carried out with Speedboat technology.
2025

2025
Sale of 51% of Creo Medical Europe completed, and launched devices SpydrBlade Flex, Speedboat Notch and Reusable Interface Cable.






